Key Releases are ad hoc announcements pursuant to SIX Swiss Exchange Article 53 Listing Rules.
Header
News archive
News Archive Navigation
icon
News Archive Navigation Language
Language Preferences
Showing 1817 results
December 2016
-
Media ReleaseNovartis LEE011 (ribociclib) plus letrozole analyses show superior PFS across broad spectrum of patients in first-line HR+/HER2- advanced breast cancer versus letrozoleMONALEESA-2 analyses demonstrate superior PFS with LEE011 plus letrozole in pre-defined patient subgroups - from de novo to bone, liver and lung metastases - compared to letrozole alone …
-
Media ReleaseNovartis' Ultibro® Breezhaler® improved lung function and COPD symptoms after direct switch from previous treatmentUltibro® Breezhaler® improved lung function and breathlessness after direct switch from long-acting bronchodilators or steroid-containing combination therapies Results further support the…
-
Media ReleaseNovartis' Lucentis® received EU approval in new indication - Lucentis the only treatment available for a wide range of CNV conditionsThe European Commission approved Lucentis to treat patients for visual impairment due to choroidal neovascularization (CNV) associated with causes other than neovascular age-related macular…
-
Media ReleaseNovartis announces Zykadia® first-line study results showing 16.6 month progression-free survival in patients with ALK+ advanced NSCLCPatients without brain metastases at diagnosis experienced median progression-free survival of 26.3 months, the longest seen in a global Phase III study in ALK+ NSCLC[1] Patients with…
-
Featured NewsHealth experts explore response to chronic illness in poor countries
Health experts from around the world met November 29 to discuss ways to tackle an emerging crisis.
-
Media ReleaseNovartis data shows Treatment-free Remission rates are consistently above 50% regardless of reason for switch to Tasigna® from Glivec®ENESTop post-hoc analysis provides further insights into Treatment-free Remission (TFR) among Ph+ CML switch patients[1] The Tasigna TFR trials, including ENESTop, demonstrate our continued…
-
Media ReleaseNovartis survey uncovers real-world burden of myeloproliferative neoplasms (rare blood cancers) on daily activity and ability to workFindings across six countries and four continents showed a reduction in quality of life for majority (71%) of patients living with myeloproliferative neoplasms (MPNs)[1] Patients…
-
Media ReleaseNew interim data demonstrate Sandoz proposed biosimilar rituximab has equivalent efficacy to reference productASSIST-FL trial demonstrates equivalent safety, efficacy, pharmacokinetics and pharmacodynamics of Sandoz proposed biosimilar rituximab (GP2013) to the reference product[1] Interim data in over…
-
Media ReleaseNovartis presents results from first global registration trial of CTL019 in pediatric and young adult patients with r/r B-ALL82% (41 of 50) of patients achieved complete remission or complete remission with incomplete blood count recovery in interim analysis of Novartis study (ELIANA) ELIANA, the first global CAR T…
-
Media ReleaseNovartis SEG101 (crizanlizumab, formerly SelG1) significantly reduces frequency of sickle cell pain crises in Phase II studySEG101 reduced annual rate of sickle cell-related pain crises (SCPC) by 45.3% compared to placebo in patients with or without hydroxyurea therapy SEG101 is a potential new disease-…
-
Story From Our Labs
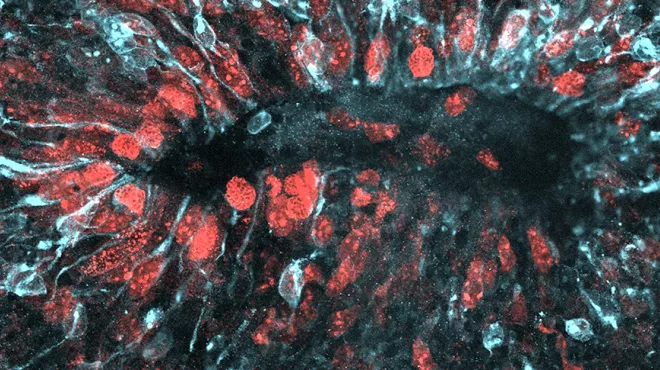 How Zika infects the growing brain
How Zika infects the growing brainThe fast-spreading virus can take multiple routes into developing human nerve cells.
-
Story Discovery
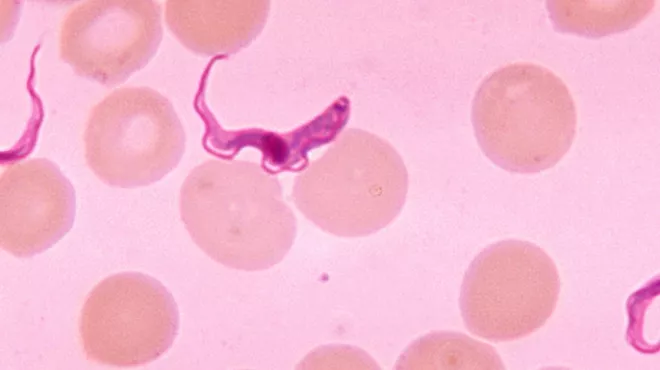 Killing three diseases with one stone in the lab
Killing three diseases with one stone in the labNovartis researchers have discovered a compound that is showing promise against three parasites that cause three neglected diseases.
Pagination
- ‹ Previous page
- 1
- …
- 115
- 116
- 117
- 118
- 119
- 120
- 121
- …
- 152
- › Next page